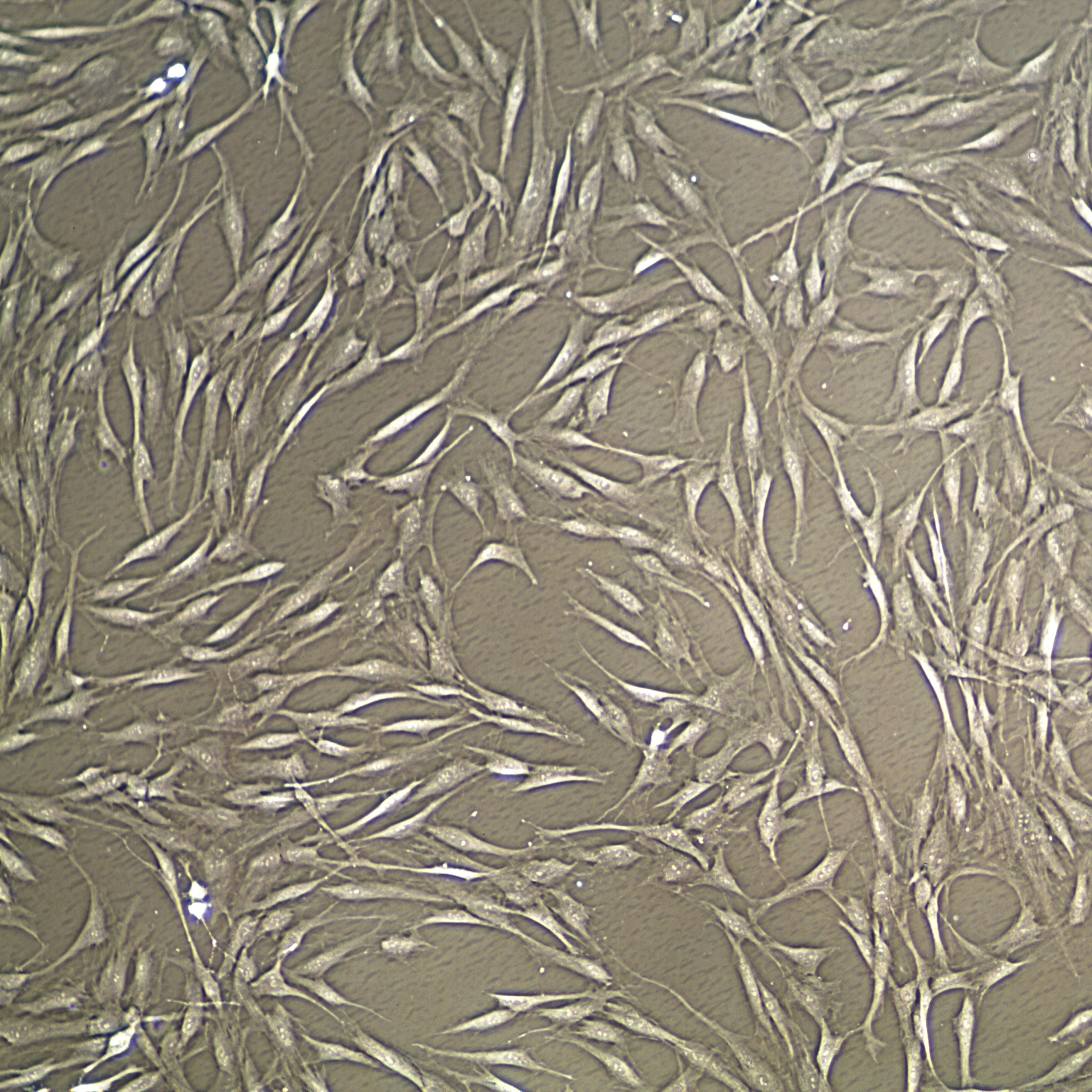
Studies have also found that functional and phenotypic differences may exist across tissue sources, culture conditions, and extent of ex-vivo expansion. Since then, additional markers have been identified and widely accepted as reliable MSC surface markers, such as STRO-1, CD271, CD348, CD200, and CD105. The basic characteristics and minimal criteria of MSCs were declared in 2006 by the International Society for Cellular Therapy (ISCT) (Fig. We adapt to your requirements by providing either individual service modules or a fully comprehensive service package.ĭue to the population heterogeneity of MSC cultures resulting from factors such as different tissue derivations, diverse isolation protocols, various growth conditions, a common guideline for MSC preparations is required to be defined to characterize MSC populations. With the help of our well-established technologies and years of experience, Creative Biolabs can provide phenotypic, genetic, and functional assessments of your MSC population of interest using various techniques and tools. This can be broken down into identity, potency, purity, and safety that must all be considered during product development. One of the most important aspects of the manufacturing of cell-based therapy products is the definition and measurement of cell characteristics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
